MTS Helps Medical Device Manufacturer Develop 3D-Printed Spinal Implants
MTS Systems Corporation, a leading global supplier of high-performance test systems, motion simulators and sensors, today announced the use of MTS material test systems to develop 3D-printed, porous titanium spinal implants.
As the world’s population ages, spinal surgery is becoming more commonplace. Along with the deterioration that comes with natural aging, spinal tumors or trauma can cause decayed, diseased or collapsed discs that are immensely painful and need to be repaired.
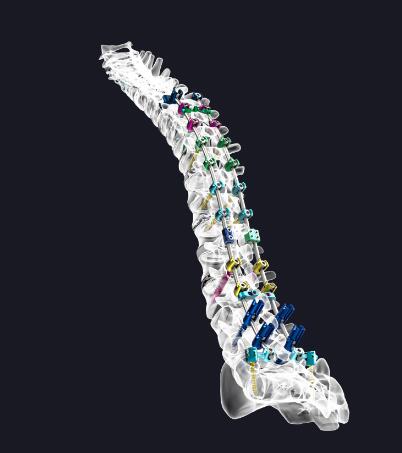 Image via: NEXXT SPINE
Image via: NEXXT SPINE
To address these concerns, Nexxt Spine, a medical device company focused on designing, manufacturing, and distributing innovative spinal solutions, is using MTS material test systems to develop bone healing spinal implants. “We’ve immersed ourselves in the additive manufacturing space completely and positioned our future business focus as pioneers in the design and development of spinal fusion implants that incorporate interlaced micro-lattice architectures with the goal of promoting osteoconduction, osteointegration and bony fusion,” says Alaedeen Abu-Mulaweh, Nexxt Spine Director of Engineering. “Given the highly nuanced nature of these intricate micro geometries, the MTS testing platform has been our go-to solution for quantifying and tailoring the associated biomechanical performance attributes from day one.”
“MTS’ expertise in creating systems for testing biomedical devices and simulating in-vivo conditions, combined with leadership in developing testing techniques for additively manufactured products, brings a unique set of knowledge to this type of testing application,” says Dr. Jeffrey Graves, MTS President and CEO. “MTS is pleased to support medical device manufacturers in their quest to design innovative products that improve health and well-being. In this case, MTS material test systems are helping Nexxt Spine find better ways to repair spines and improve quality of life for spine surgery patients.”
Source:MTS



Recent Comments