Oxford Performance Materials and JSR Corporation Announce Partnership to Advance OPM Technology Platform Throughout Asia
Oxford Performance Materials, Inc. (OPM), a leader in advanced materials science and high performance additive manufacturing (HPAM®), and Tokyo-based JSR Corporation (JSR), a leading materials supplier in a variety of technology driven markets, today announced a partnership to advance OPM’s poly-ether-ketone-ketone (PEKK)-based 3D-printed orthopedic devices and biomedical & dental materials businesses throughout Asia.
Under the terms of the agreement, OPM and JSR will establish operations in Japan as OPM Asia, with an exclusive regional license to exploit OPM’s proprietary OsteoFab® biomedical device 3D printing technology and OXPEKK® materials technology. JSR will be providing the equity capital for OPM Asia that will enable the establishment of a world-class biomedical manufacturing and market development operation. The OPM Asia license will cover a broad territory consisting of Japan, China, Taiwan, Korea, Brunei Darussalam, Cambodia, Lao PDR, Malaysia, Myanmar, Philippines, Singapore, Thailand, Vietnam and India.

“Asia’s healthcare industry is growing rapidly, and with that growth comes increased demand for medical innovation that delivers improved clinical effectiveness to more people with superior economics,” said Scott DeFelice, Chief Executive Officer of Oxford Performance Materials. The healthcare industry in Asia is expected to grow by 11.1% to $517 billion in 2018, representing one of the fastest growing regions in the world, according to Frost & Sullivan. OPM has long identified Asia as an attractive market due to clear demographic trends and a predictable regulatory and reimbursement environment.
JSR has a rapidly growing life sciences business and broad strategic partnerships across Asia. Regarding the OPM Asia partnership, DeFelice stated, “JSR has built an impressive personalized medicine ecosystem, with capabilities in medical 3D software, drug discovery & delivery, and biologics. JSR’s regulatory expertise and partnerships with healthcare providers, including joint development of the JSR-Keio University Medical and Chemical Innovation Center (JKiC), are unique in the region, and JSR will be a strong, influential partner to OPM. We are extremely pleased that JSR has decided to finance the OPM Asia enterprise and work with us to establish the OPM technology platform throughout Japan and the wider region.”
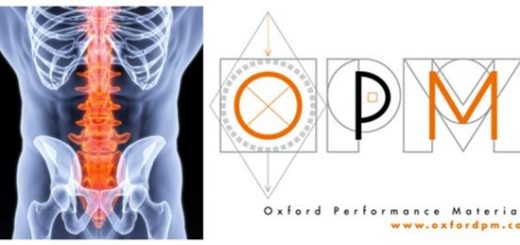
The global adoption of OPM’s 3D printed PEKK-based medical implants over metallic and other polymeric options in craniomaxillofacial (CMF), spine and other emerging orthopedic and neurologic indications is being driven by a combination of OsteoFab 3D printing technology attributes, including desirable economics, biocompatibility, radiolucency, and bone-like mechanics and behavior. Studies have also shown that OPM’s OsteoFab surface technology exhibits antibacterial characteristics, inhibiting bacterial attachment, as well as osteoconductive properties – meaning bony tissue grows onto the surface of OsteoFab implants. These are two critical attributes that may improve patient recovery and long-term implant fixation. OPM also has a robust biomedical materials business, manufacturing OXPEKK formulations of semi-finished goods for medical and dental implant applications.
As previously announced, OPM was granted Accreditation of Foreign Medical Device Manufacturer by the Japanese Ministry of Health, Labour and Welfare in March 2018. This accreditation helps lay the groundwork for OPM’s expansion into the rapidly growing Asian healthcare market. OPM Asia is scheduled to establish its Japanese manufacturing facility in late 2018 with products commercially available to the Asian market in Q2, 2019.
Scott DeFelice will be CEO of OPM Asia and serve on its Board of Directors. Joining Mr. DeFelice on the founding board will be Mr. Bernard Plishtin from OPM and one member from JSR
Source: .OPM


Recent Comments